111 19.4 Parasitic Infections of the Circulatory and Lymphatic Systems
Learning Objectives
- Identify common parasites that cause infections of the circulatory and lymphatic systems
- Compare the major characteristics of specific parasitic diseases affecting the circulatory and lymphatic systems
Some protozoa and parasitic flukes are also capable of causing infections of the human circulatory system. Although these infections are rare in the US, they continue to cause widespread suffering in the developing world today. Fungal infections of the circulatory system are very rare. Therefore, they are not discussed in this chapter.
Malaria
Despite more than a century of intense research and clinical advancements, malaria remains one of the most important infectious diseases in the world today. Its widespread distribution places more than half of the world’s population in jeopardy. In 2015, the WHO estimated there were about 214 million cases of malaria worldwide, resulting in about 438,000 deaths; about 88% of cases and 91% of deaths occurred in Africa.[1] Although malaria is not currently a major threat in the US, the possibility of its reintroduction is a concern. Malaria is caused by several protozoan parasites in the genus Plasmodium: P. falciparum, P. knowlesi, P. malariae, P. ovale, and P. vivax. Plasmodium primarily infect red blood cells and are transmitted through the bite of Anopheles mosquitoes.
Currently, P. falciparum is the most common and most lethal cause of malaria, often called falciparum malaria. Falciparum malaria is widespread in highly populated regions of Africa and Asia, putting many people at risk for the most severe form of the disease.
The classic signs and symptoms of malaria are cycles of extreme fever and chills. The sudden, violent symptoms of malaria start with malaise, abrupt chills, and fever (39–41° C [102.2–105.8 °F]), rapid and faint pulse, polyuria, headache, myalgia, nausea, and vomiting. After 2 to 6 hours of these symptoms, the fever falls, and profuse sweating occurs for 2 to 3 hours, followed by extreme fatigue. These symptoms are a result of Plasmodium emerging from red blood cells synchronously, leading to simultaneous rupture of a large number of red blood cells, resulting in damage to the spleen, liver, lymph nodes, and bone marrow. The organ damage resulting from hemolysis causes patients to develop sludge blood (i.e., blood in which the red blood cells agglutinate into clumps) that can lead to lack of oxygen, necrosis of blood vessels, organ failure, and death.
In established infections, malarial cycles of fever and chills typically occur every 2 days in the disease described as tertian malaria, which is caused by P. vivax and P. ovale. The cycles occur every 3 days in the disease described as quartan malaria, which is caused by P. malariae. These intervals may vary among cases.
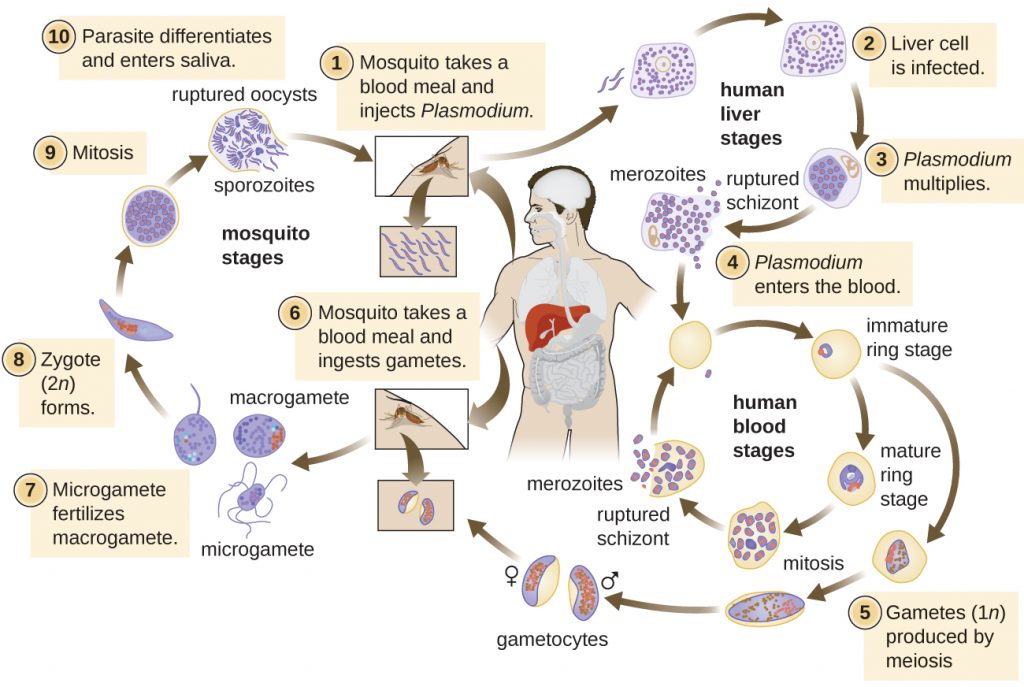
Plasmodium has a complex life cycle that includes several developmental stages alternately produced in mosquitoes and humans (Figure 19.24). When an infected mosquito takes a blood meal, sporozoites in the mosquito salivary gland are injected into the host’s blood. These parasites circulate to the liver, where they develop into schizonts. The schizonts then undergo schizogony, resulting in the release of many merozoites at once. The merozoites move to the bloodstream and infect red blood cells. Inside red blood cells, merozoites develop into trophozoites that produce more merozoites. The synchronous release of merozoites from red blood cells in the evening leads to the symptoms of malaria.
In addition, some trophozoites alternatively develop into male and female gametocytes. The gametocytes are taken up when the mosquito takes a blood meal from an infected individual. Sexual sporogony occurs in the gut of the mosquito. The gametocytes fuse to form zygotes in the insect gut. The zygotes become motile and elongate into an ookinete. This form penetrates the midgut wall and develops into an oocyst. Finally, the oocyst releases new sporozoites that migrate to the mosquito salivary glands to complete the life cycle.
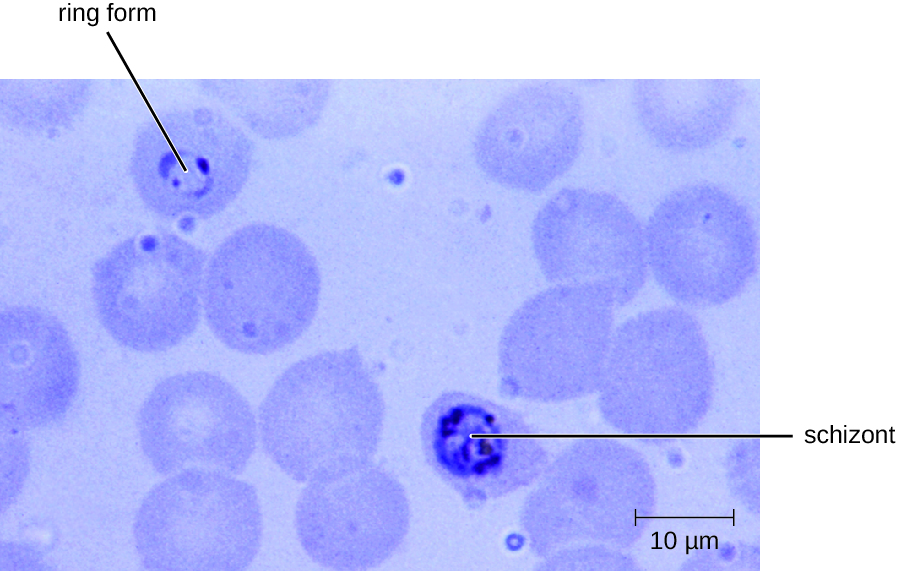
Diagnosis of malaria is by microscopic observation of developmental forms of Plasmodium in blood smears and rapid EIA assays that detect Plasmodium antigens or enzymes (Figure 19.25). Drugs such as chloroquine, atovaquone , artemether, and lumefantrine may be prescribed for both acute and prophylactic therapy, although some Plasmodium spp. have shown resistance to antimalarial drugs. Use of insecticides and insecticide-treated bed nets can limit the spread of malaria. Despite efforts to develop a vaccine for malaria, none is currently available.

Visit this site to learn how the parasite Plasmodium infects red blood cells.
The Nothing But Nets campaign, an initiative of the United Nations Foundation, has partnered with the Bill and Melinda Gates Foundation to make mosquito bed nets available in developing countries in Africa. Visit their website to learn more about their efforts to prevent malaria.
- Why is malaria one of the most important infectious diseases?
Toxoplasmosis
The disease toxoplasmosis is caused by the protozoan Toxoplasma gondii. T. gondii is found in a wide variety of birds and mammals,[2] and human infections are common. The Centers for Disease Control and Prevention (CDC) estimates that 22.5% of the population 12 years and older has been infected with T. gondii; but immunocompetent individuals are typically asymptomatic, however.[3] Domestic cats are the only known definitive hosts for the sexual stages of T. gondii and, thus, are the main reservoirs of infection. Infected cats shed T. gondii oocysts in their faeces, and these oocysts typically spread to humans through contact with faecal matter on cats’ bodies, in litter boxes, or in garden beds where outdoor cats defecate.
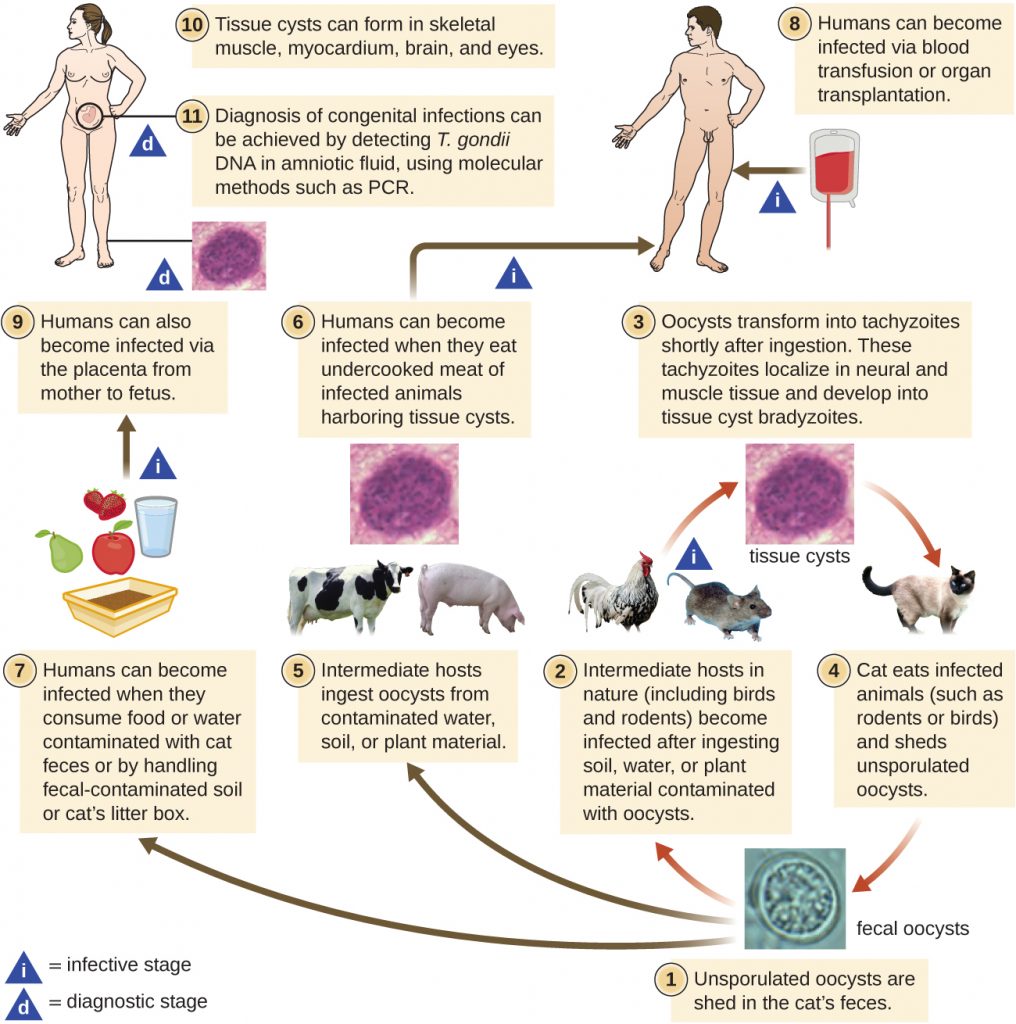
T. gondii has a complex life cycle that involves multiple hosts. The T. gondii life cycle begins when unsporulated oocysts are shed in the cat’s feces. These oocysts take 1–5 days to sporulate in the environment and become infective. Intermediate hosts in nature include birds and rodents, which become infected after ingesting soil, water, or plant material contaminated with the infective oocysts. Once ingested, the oocysts transform into tachyzoites that localize in the bird or rodent neural and muscle tissue, where they develop into tissue cysts. Cats may become infected after consuming birds and rodents harboring tissue cysts. Cats and other animals may also become infected directly by ingestion of sporulated oocysts in the environment. Interestingly, Toxoplasma infection appears to be able to modify the host’s behavior. Mice infected by Toxoplasma lose their fear of cat pheromones. As a result, they become easier prey for cats, facilitating the transmission of the parasite to the cat definitive host[4] (Figure 19.26).
Toxoplasma infections in humans are extremely common, but most infected people are asymptomatic or have subclinical symptoms. Some studies suggest that the parasite may be able to influence the personality and psychomotor performance of infected humans, similar to the way it modifies behavior in other mammals.[5] When symptoms do occur, they tend to be mild and similar to those of mononucleosis. However, asymptomatic toxoplasmosis can become problematic in certain situations. Cysts can lodge in a variety of human tissues and lie dormant for years. Reactivation of these quiescent infections can occur in immunocompromised patients following transplantation, cancer therapy, or the development of an immune disorder such as AIDS. In patients with AIDS who have toxoplasmosis, the immune system cannot combat the growth of T. gondii in body tissues; as a result, these cysts can cause encephalitis, retinitis, pneumonitis, cognitive disorders, and seizures that can eventually be fatal.
Toxoplasmosis can also pose a risk during pregnancy because tachyzoites can cross the placenta and cause serious infections in the developing fetus. The extent of fetal damage resulting from toxoplasmosis depends on the severity of maternal disease, the damage to the placenta, the gestational age of the fetus when infected, and the virulence of the organism. Congenital toxoplasmosis often leads to fetal loss or premature birth and can result in damage to the central nervous system, manifesting as mental retardation, deafness, or blindness. Consequently, pregnant women are advised by the CDC to take particular care in preparing meat, gardening, and caring for pet cats.[6] Diagnosis of toxoplasmosis infection during pregnancy is usually achieved by serology including TORCH testing (the “T” in TORCH stands for toxoplasmosis). Diagnosis of congenital infections can also be achieved by detecting T. gondii DNA in amniotic fluid, using molecular methods such as PCR.
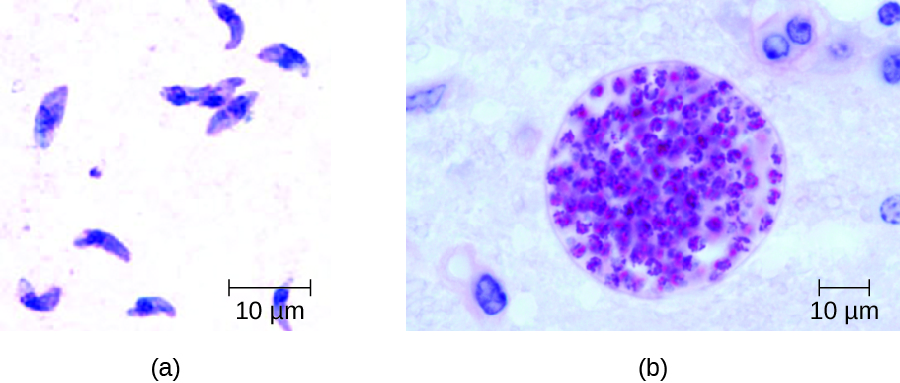
In adults, diagnosis of toxoplasmosis can include observation of tissue cysts in tissue specimens. Tissue cysts may be observed in Giemsa- or Wright-stained biopsy specimens, and CT, magnetic resonance imaging, and lumbar puncture can also be used to confirm infection (Figure 19.27).
Preventing infection is the best first-line defense against toxoplasmosis. Preventive measures include washing hands thoroughly after handling raw meat, soil, or cat litter, and avoiding consumption of vegetables possibly contaminated with cat feces. All meat should be cooked to an internal temperature of 73.9–76.7 °C (165–170 °F).
Most immunocompetent patients do not require clinical intervention for Toxoplasma infections. However, neonates, pregnant women, and immunocompromised patients can be treated with pyrimethamine and sulfadiazine—except during the first trimester of pregnancy, because these drugs can cause birth defects. Spiramycin has been used safely to reduce transmission in pregnant women with primary infection during the first trimester because it does not cross the placenta.
- How does T. gondii infect humans?
Babesiosis
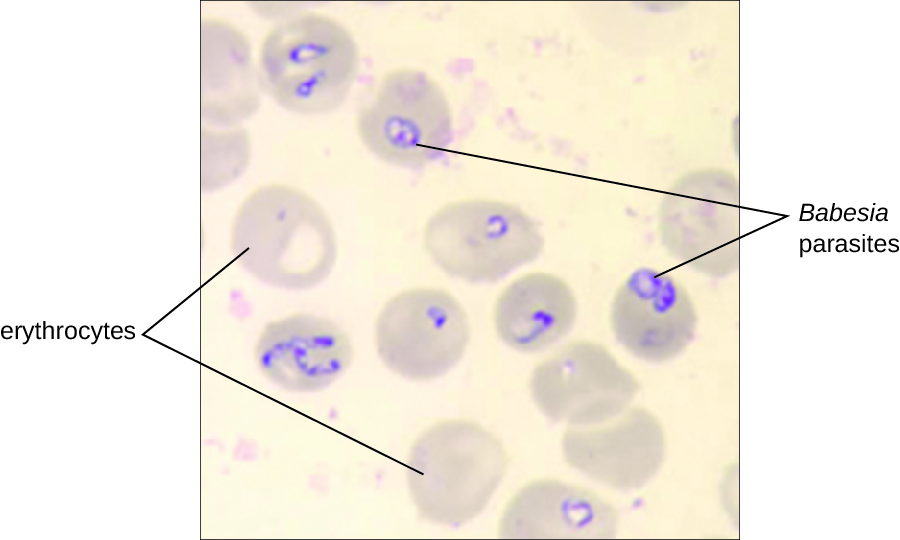
Babesiosis is a rare zoonotic infectious disease caused by Babesia spp. These parasitic protozoans infect various wild and domestic animals and can be transmitted to humans by black-legged Ixodes ticks. In humans, Babesia infect red blood cells and replicate inside the cell until it ruptures. The Babesia released from the ruptured red blood cell continue the growth cycle by invading other red blood cells. Patients may be asymptomatic, but those who do have symptoms often initially experience malaise, fatigue, chills, fever, headache, myalgia, and arthralgia. In rare cases, particularly in asplenic (absence of the spleen) patients, the elderly, and patients with AIDS, babesiosis may resemble falciparum malaria, with high fever, hemolytic anaemia, hemoglobinuria (hemoglobin or blood in urine), jaundice, and renal failure, and the infection can be fatal. Previously acquired asymptomatic Babesia infection may become symptomatic if a splenectomy is performed.
Diagnosis is based mainly on the microscopic observation of parasites in blood smears (Figure 19.285). Serologic and antibody detection by IFA can also be performed and PCR-based tests are available. Many people do not require clinical intervention for Babesia infections, however, serious infections can be cleared with a combination of atovaquone and azithromycin or a combination of clindamycin and quinine.
Chagas Disease
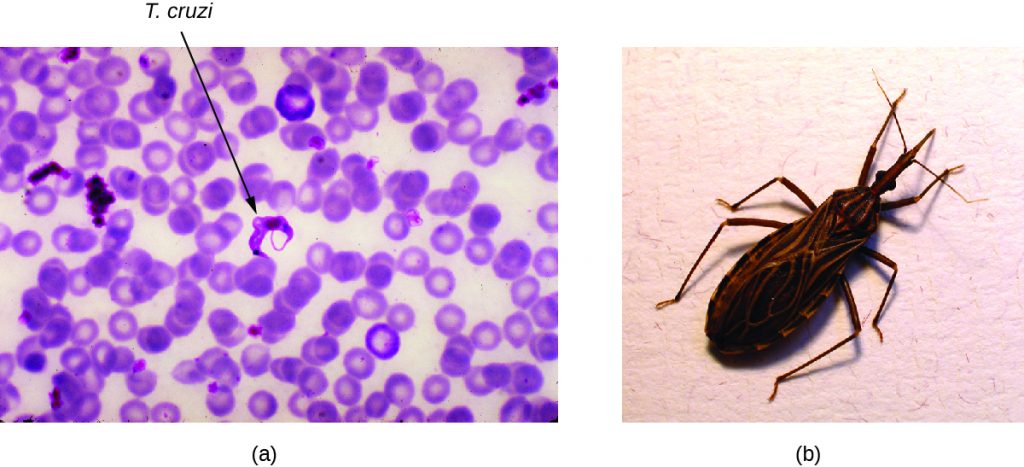
Also called American trypanosomiasis, Chagas disease is a zoonosis classified as a neglected tropical disease (NTD). It is caused by the flagellated protozoan Trypanosoma cruzi and is most commonly transmitted to animals and people through the feces of triatomine bugs. The triatomine bug is nicknamed the kissing bug because it frequently bites humans on the face or around the eyes; the insect often defecates near the bite and the infected fecal matter may be rubbed into the bite wound by the bitten individual (Figure 19.29). The bite itself is painless and, initially, many people show no signs of the disease. Alternative modes of transmission include contaminated blood transfusions, organ transplants from infected donors, and congenital transmission from mother to fetus.
Chagas disease is endemic throughout much of Mexico, Central America, and South America, where, according to WHO, an estimated 6 million to 7 million people are infected.[7] Currently, Chagas disease is not endemic in the US, even though triatomine bugs are found in the southern half of the country.
Triatomine bugs typically are active at night, when they take blood meals by biting the faces and lips of people or animals as they sleep and often defecate near the site of the bite. Infection occurs when the host rubs the feces into their eyes, mouth, the bite wound, or another break in the skin. The protozoan then enters the blood and invades tissues of the heart and central nervous system, as well as macrophages and monocytes. Nonhuman reservoirs of T. cruzi parasites include wild animals and domesticated animals such as dogs and cats, which also act as reservoirs of the pathogen.[8]
There are three phases of Chagas disease: acute, intermediate, and chronic. These phases can be either asymptomatic or life-threatening depending on the immunocompetence status of the patient.
In acute phase disease, symptoms include fever, headache, myalgia, rash, vomiting, diarrhea, and enlarged spleen, liver, and lymph nodes. In addition, a localized nodule called a chagoma may form at the portal of entry, and swelling of the eyelids or the side of the face, called Romaña’s sign, may occur near the bite wound. Symptoms of the acute phase may resolve spontaneously, but if untreated, the infection can persist in tissues, causing irreversible damage to the heart or brain. In rare cases, young children may die of myocarditis or meningoencephalitis during the acute phase of Chagas disease.
Following the acute phase is a prolonged intermediate phase during which few or no parasites are found in the blood and most people are asymptomatic. Many patients will remain asymptomatic for life; however, decades after exposure, an estimated 20%–30% of infected people will develop chronic disease that can be debilitating and sometimes life threatening. In the chronic phase, patients may develop painful swelling of the colon, leading to severe twisting, constipation, and bowel obstruction; painful swelling of the esophagus, leading to dysphagia and malnutrition; and flaccid cardiomegaly (enlargement of the heart), which can lead to heart failure and sudden death.
Diagnosis can be confirmed through several different tests, including direct microscopic observation of trypanosomes in the blood, IFA, EIAs, PCR, and culturing in artificial media. In endemic regions, xenodiagnoses may be used; this method involves allowing uninfected kissing bugs to feed on the patient and then examining their feces for the presence of T. cruzi.
The medications nifurtimox and benznidazole are effective treatments during the acute phase of Chagas disease. The efficacy of these drugs is much lower when the disease is in the chronic phase. Avoiding exposure to the pathogen through vector control is the most effective method of limiting this disease.
- How do kissing bugs infect humans with Trypanosoma cruzi?
Leishmaniasis
Although it is classified as an NTD, leishmaniasis is relatively widespread in tropical and subtropical regions, affecting people in more than 90 countries. It is caused by approximately 20 different species of Leishmania, protozoan parasites that are transmitted by sand fly vectors such as Phlebotomus spp. and Lutzomyia spp. Dogs, cats, sheep, horses, cattle rodents, and humans can all serve as reservoirs.
The Leishmania protozoan is phagocytosed by macrophages but uses virulence factors to avoid destruction within the phagolysosome. The virulence factors inhibit the phagolysosome enzymes that would otherwise destroy the parasite. The parasite reproduces within the macrophage, lyses it, and the progeny infect new macrophages (see When Phagocytosis Fails).
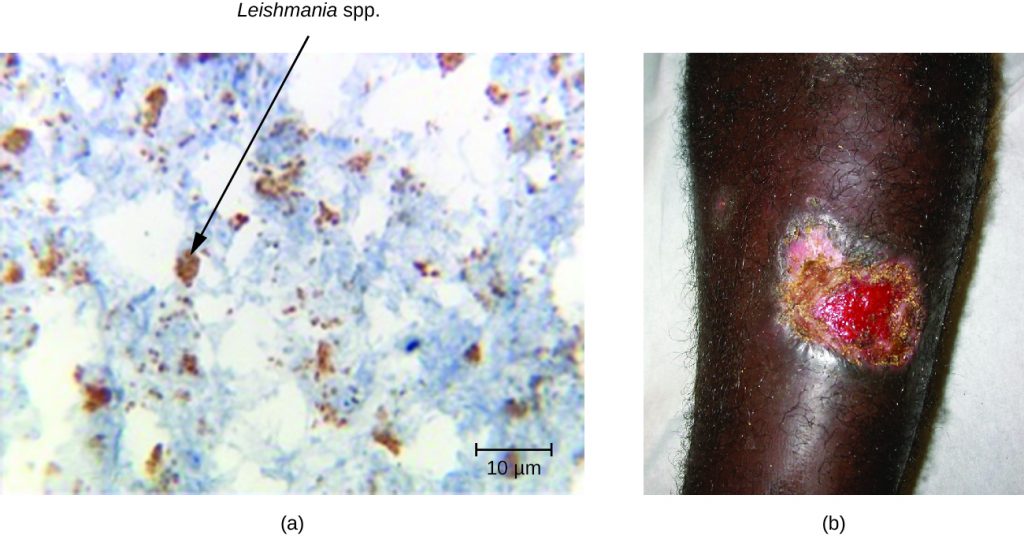
The three major clinical forms of leishmaniasis are cutaneous (oriental sore, Delhi boil, Aleppo boil), visceral (kala-azar, Dumdum fever), and mucosal (espundia). The most common form of disease is cutaneous leishmaniasis, which is characterized by the formation of sores at the site of the insect bite that may start out as papules or nodules before becoming large ulcers (Figure 19.30).
It may take visceral leishmaniasis months and sometimes years to develop, leading to enlargement of the lymph nodes, liver, spleen, and bone marrow. The damage to these body sites triggers fever, weight loss, and swelling of the spleen and liver. It also causes a decrease in the number of red blood cells (anaemia), white blood cells (leukopenia), and platelets (thrombocytopenia), causing the patient to become immunocompromised and more susceptible to fatal infections of the lungs and gastrointestinal tract.
The mucosal form of leishmaniasis is one of the less common forms of the disease. It causes a lesion similar to the cutaneous form but mucosal leishmaniasis is associated with mucous membranes of the mouth, nares, or pharynx, and can be destructive and disfiguring. Mucosal leishmaniasis occurs less frequently when the original cutaneous (skin) infection is promptly treated.
Definitive diagnosis of leishmaniasis is made by visualizing organisms in Giemsa-stained smears, by isolating Leishmania protozoans in cultures, or by PCR-based assays of aspirates from infected tissues. Specific DNA probes or analysis of cultured parasites can help to distinguish Leishmania species that are causing simple cutaneous leishmaniasis from those capable of causing mucosal leishmaniasis.
Cutaneous leishmaniasis is usually not treated. The lesions will resolve after weeks (or several months), but may result in scarring. Recurrence rates are low for this disease. More serious infections can be treated with stibogluconate (antimony gluconate), amphotericin B, and miltefosine.
- Compare the mucosal and cutaneous forms of leishmaniasis.
DISEASE PROFILE: Common Protozoal Infections of the Human Circulatory System
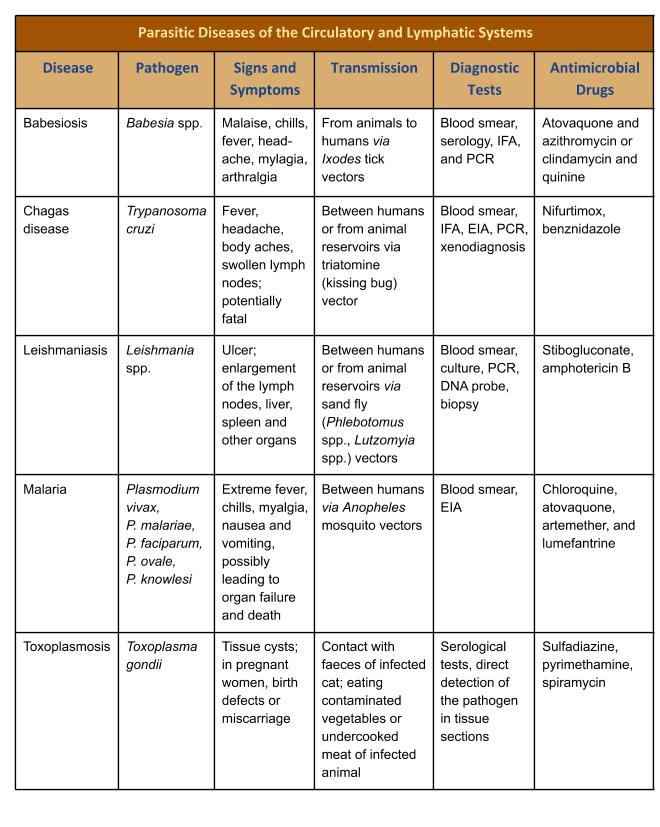
Table 19.3. Parasitic Diseases of the Circulatory and Lymphatic Systems
CLINICAL FOCUS: Resolution
Despite continued antibiotic treatment and the removal of the venous catheter, Barbara’s condition further declined. She began to show signs of shock and her blood pressure dropped to 77/50 mmHg. Anti-inflammatory drugs and drotrecogin-α were administered to combat sepsis. However, by the seventh day of hospitalization, Barbara experienced hepatic and renal failure and died.
Staphylococcus aureus most likely formed a biofilm on the surface of Barbara’s catheter. From there, the bacteria were chronically shed into her circulation and produced the initial clinical symptoms. The chemotherapeutic therapies failed in large part because of the drug-resistant MRSA isolate. Virulence factors like leukocidin and hemolysins also interfered with her immune response. Barbara’s ultimate decline may have been a consequence of the production of enterotoxins and toxic shock syndrome toxin (TSST), which can initiate toxic shock.
Venous catheters are common life-saving interventions for many patients requiring long-term administration of medication or fluids. However, they are also common sites of bloodstream infections. According to the CDC, in the US alone, thousands of people die annually from catheter-related bloodstream infections.[9]
Go back to the previous Clinical Focus box.
Key Takeaways
- Malaria is a protozoan parasite that remains an important cause of death primarily in the tropics. Several species in the genus Plasmodium are responsible for malaria and all are transmitted by Anopheles mosquitoes. Plasmodium infects and destroys human red blood cells, leading to organ damage, anaemia, blood vessel necrosis, and death. Malaria can be treated with various antimalarial drugs and prevented through vector control.
- Toxoplasmosis is a widespread protozoal infection that can cause serious infections in the immunocompromised and in developing fetuses. Domestic cats are the definitive host.
- Babesiosis is a generally asymptomatic infection of red blood cells that can causes malaria-like symptoms in elderly, immunocompromised, or asplenic patients.
- Chagas disease is a tropical disease transmitted by triatomine bugs. The trypanosome infects heart, neural tissues, monocytes, and phagocytes, often remaining latent for many years before causing serious and sometimes fatal damage to the digestive system and heart.
- Leishmaniasis is caused by the protozoan Leishmania and is transmitted by sand flies. Symptoms are generally mild, but serious cases may cause organ damage, anaemia, and loss of immune competence.
Additional Resources
Multiple Choice
Fill in the Blank
Short Answer
- Describe main cause of Plasmodium falciparum infection symptoms.
- Why should pregnant women avoid cleaning their cat’s litter box or do so with protective gloves?
Critical Thinking
- What measures can be taken to reduce the likelihood of malaria reemerging in the US?
- World Health Organization. “World Malaria Report 2015: Summary.” 2015. http://www.who.int/malaria/publications/world-malaria-report-2015/report/en/. Accessed May 14, 2019. ↵
- A.M. Tenter et al.. “Toxoplasma gondii: From Animals to Humans.” International Journal for Parasitology 30 no. 12-13 (2000):1217–1258. ↵
- Centers for Disease Control and Prevention. “Parasites - Toxoplasmosis (Toxoplasma Infection). Epidemiology & Risk Factors.” 2015 http://www.cdc.gov/parasites/toxoplasmosis/epi.html. Accessed May 14, 2019. ↵
- J. Flegr. “Effects of Toxoplasma on Human Behavior.” Schizophrenia Bulletin 33, no. 3 (2007):757–760. ↵
- Ibid. ↵
- Centers for Disease Control and Prevention. “Parasites - Toxoplasmosis (Toxoplasma infection). Toxoplasmosis Frequently Asked Questions (FAQs).” 2013. http://www.cdc.gov/parasites/toxoplasmosis/gen_info/faqs.html. Accessed May 14, 2019. ↵
- World Health Organization. “Chagas disease (American trypanosomiasis). Fact Sheet.” 2016. http://www.who.int/mediacentre/factsheets/fs340/en/. Accessed May 14, 2019. ↵
- C.E. Reisenman et al. “Infection of Kissing Bugs With Trypanosoma cruzi, Tucson, Arizona, USA.” Emerging Infectious Diseases 16 no. 3 (2010):400–405. ↵
- Centers for Disease Control and Prevention. “Central Line-associated Bloodstream Infection (CLABSI)” 2016. https://www.cdc.gov/hai/bsi/bsi.html. Accessed May 14, 2019. ↵
